Silver (Ag) and Copper (Cu) were used in the time of the Persian monarchs for water disinfection and sanitisation, as well as for food preservation (Lemire et al, 2013). Silver foils have been used to prevent surgical wound infection, while copper compounds have been widely used in agriculture as fungicides on grapes and potatoes (Lemire et al, 2013). Copper was also prescribed for respiratory diseases and used for purifying water in ancient Greece (400BC; Dollwet and Sorenson, 2001). Copper was acknowledged as the first metal with antibacterial properties by the United States Environmental Protection Agency (US EPA) in 2008 (Pradot al, 2012). Copper is considerably cheaper and more easily accessible compared with silver and gold. However, the native form of copper ions is commonly converted into nanoparticles in order to optimise the efficacy of copper in wound healing (Tiwari et al, 2014). Copper nanoparticles (CuNPs) have potentially enhanced catalytic activity and improved bioavailability in comparison to the native form (Tiwari et al, 2014). In addition, copper nanoparticles that exhibit biocompatibility can be manufactured using eco-friendly processes.
Nanotechnology is a novel therapy in wound management that incorporates nanometer-sized materials, with implications for tissue repair, regenerative health and disease prevention (Zarrintaj et al, 2017). In addition to healthcare or wound management, copper-based nanoparticles have also been used in various ways, including as an antimicrobial coating for dental hygiene and virus disinfection. The use of nanoparticles provide a new, alternative to aid the wound healing process. Furthermore, it has unique properties as a bactericidal agent (Barroso et al, 2020).
In view of the antimicrobial abilities and low toxicity properties, metal nanoparticles comprised of copper, silver, gold and zinc are ideal for incorporation into wound dressings (Negut et al, 2018). Copper has a low level of resistance among microbes that are clinically significant, which is one of the key advantages of using it as a bactericidal agent. It also has a complex role in various tissue cells where it modulates various cytokines and growth factors that are an essential component in all stages of wound healing (Kornblatt et al, 2016) The bactericidal activity of copper is mainly attributed to the release of ions, which affects the integrity of the membrane and the bacterial cell wall, generates intracellular oxidative stress and is genotoxic, resulting in the death of microorganisms (Grass et al, 2010; Vincent et al, 2018). In general, CuNPs have a greater ability to engage and interact with the membrane of bacteria and subsequently compromise the structural integrity of bacterial membranes.
Intact skin functions as a protective barrier that effectively prevents microbial invasion. Any break in the skin integrity and compromise to the epidermal barrier will facilitate bacteria to enter the deeper layers of the skin with a higher risk of infection. All open wounds are vulnerable to contamination and microbial colonisation, although not all contaminated wounds will develop an infection (International Wound Infection Institute, 2022). Microbial infection remains a major factor in delayed wound healing in chronic hard-to-heal wounds. Other factors include hypoxia or poor perfusion; repetitive ischaemia-reperfusion injury; and inadequate offloading or compression therapy (Malone et al, 2017). The Wound Healing Society categorises chronic wounds into four different groups based on the causative aetiologies, which include pressure injury, diabetic foot ulcers (DFUs), venous leg ulcers (VLUs) and arterial insufficiency ulcer (Kirsner, 2016).
Chronic wounds pose a significant global issue to the healthcare system because of their rising prevalence and substantial management costs. According to Gottrup et al (2004), it is estimated that 1–2 % of the population would suffer from a chronic wound during their lifetime in developed countries.
Biofilms play a significant role in impeding the process of wound healing and increasing susceptibility to wound infection, especially in the case of chronic wounds. Biofilms have been reported to be present in 60% (James et al, 2008) to 80% (Romling et al, 2012) of chronic wounds, and a recent meta-analysis suggests their presence is in at least 78.2% of chronic, hard-to-heal wounds (Malone et al, 2017). Therefore, early detection and identification of bioburden in wounds, and subsequent intervention to eradicate infection, is beneficial from both a clinical and economic aspect, thereby reduce burden on patients, caregivers and healthcare systems.
This case study aims to establish the clinical effects of using the cleansing agent, copper nanoparticles-infused solution, containing 0.1% ionized copper, on bacterial burden in lower leg wounds.
Materials and methods
Patients were recruited and followed up at the Wound Care Unit, Hospital Kuala Lumpur. Random sampling of patients was done among patients with wounds at the lower extremity. A variety of clinical cases were included, such as DFUs, VLUs and chronic, hard-to-heal wounds. Wound assessment was completed using the T.I.M.E. framework and bioburden parameters were assessed with MolecuLight pre- and post- wound cleansing with copper nanoparticles-infused solution.
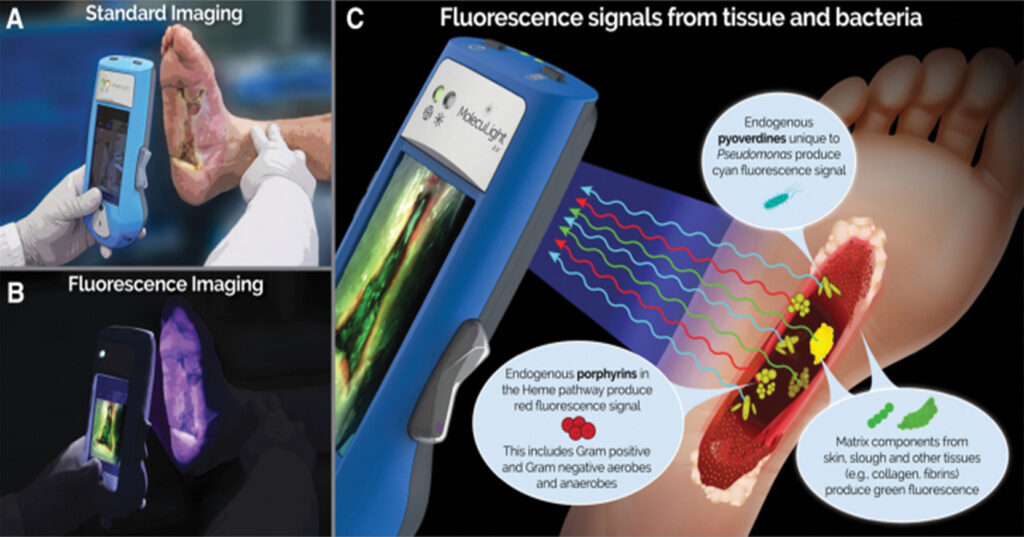
MolecuLight is a non-invasive fluorescence imaging device (Figure 1) that is able to visualise both wound tissue and bacterial fluorescence. The application of an excitation light (405nm violet light) to the wound bed tissues results in the generation of a range of fluorescence signals, and is able to create a map of bacteria in the wound bed, which is used for targeted cleansing and debridement. This imaging device requires a dark environment; therefore, all wound photos were taken under an attachable drape without ambient light exposure, to avoid misinterpretation of fluorescence or false positive results. Table 1 shows how to interpret the different fluorescent signals.
Each study subject received the same wound cleansing protocol and was followed by the standard wound treatment as practiced in the Wound Care Clinic, Hospital Kuala Lumpur.
Wound cleansing protocol
- All previous wound dressings were removed at the affected limb
- Wound assessment was completed with the T.I.M.E. framework
- MolecuLight was used over the wound bed prior to wound cleansing to identify the level of wound bioburden
- Wounds were rinsed and soaked with gauze containing copper nanoparticles-infused solution (CuHEAL++) for 10–15 minutes
- Wound bioburden was reassessed with MolecuLight after wound soaking and cleansing
- All wounds were followed by a standard of wound management, with debridement and modern wound dressings.
All patients were informed about the study, regarding the application of the nano-copper solution as the cleansing solution, and possibility of adverse effects of copper usage was explained. Furthermore, the patients consented to the use of their data and images for publications and educational use.
Results
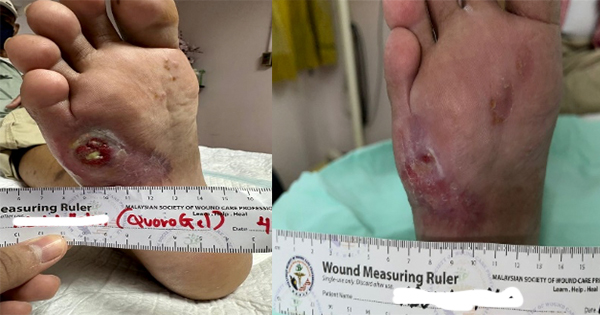
There were a total of 10 patients with wounds of the lower extremities recruited in this study, five VLUs, four DFUs and one chronic hard-to-heal wound. All wounds were assessed for bacterial bioburden pre-cleansing with nano-copper solution (CuHEAL++). MolecuLight imaging revealed positive findings in all cases and indicated a high level of bacterial bioburden in the wound bed and periwound skin area before wound cleansing.
All fluorescence images captured before wound cleansing demonstrated mostly cyan fluorescence (denoted by yellow arrow in each case) at the wound edge or periwound in eight of the cases. The other two cases showed the presence of red florescence (denoted by the white arrows) in the wound bed and wound edge.
These studies show that there is a reduction in bacterial load after using nano-copper solution as a wound cleansing agent, as evidenced by the MolecuLight fluorescence signals. Comparison between pre- and post-wound cleansing in all above clinical cases is shown in Case 1–10.
No adverse side effects were reported by any of the patients. There were no reports of copper toxicity, or dermatologic sensitivity in all clinical cases.
Discussion
The essential aspect of wound healing involves identification and subsequent reduction or elimination of bacterial bioburden. Significant bacterial loads usually demand a bacterial-targeted wound treatment procedure, such as wound cleansing, debridement or the use of antimicrobial dressings to optimise the wound bed and facilitate wound healing (Edwards and Harding, 2004). Nanoparticles that have antimicrobial properties and low toxicity to cell tissues are ideal to be used in wound management, as a cleansing solution or dressing product (Pang et al, 2023).
In the above case studies, we have shown the effectiveness of the copper nanoparticles infused solution in reducing wound bioburden over the wound bed and periwound skin area after soaking and cleansing the wound before standard wound care. Copper nanoparticles are well-recognised nanoparticles that have a broad-spectrum antimicrobial action (Sandoval et al, 2022). Copper biocidal effect is mediated through altering bacterial proteins, inhibiting the biological activity, permeabilisation of plasma membranes and membrane lipid peroxidation, which subsequently lead to bacteria cell death (Borkow and Gabbay, 2005). Besides, it also plays an important role in wound healing by promoting the development of extracellular matrix (ECM) components such as integrins, the key modifiers of cell attachment to the ECM, fibrinogen and collagen synthesis (Sandoval et al, 2022).
Copper usage is relatively safe although it has a strong biocidal action, because it is an essential trace metal that is efficiently metabolised by the human body when absorbed through tissue in comparison to silver (Melamed et al, 2021). Therefore, copper is suitable to be used as an alternative to silver and as a wound cleansing solution or incorporated into a dressing to reduce biocontamination.
Limitations
First and foremost, this is an observational study with a limited number of clinical cases that are focused on wounds of the lower extremities. A more robust study with larger number of patients and comparison of numerous clinical cases involving other part of the body (including upper extremities, trunk, sacral and gluteal) with comparison to other antiseptic solutions may be useful to support the literature evidence and validate the study’s results.
Secondly, MolecuLight has limited sensitivity in identifying wound bioburden, as it is only able to detect certain bacteria such as Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa.
Lastly, our center does not routinely perform swab or tissue culture sampling for patients unless indicated. Therefore, there is no microbiological profile taken at the same settings, in comparison to the MolecuLight results, to confirm the presence of bacteria in the wound bed in this study.
Conclusion
This case series demonstrated the clinical effectiveness and efficacy of nano-copper solution as an antimicrobial wound cleanser, which significantly reduces wound bioburden and promotes wound healing of chronic wounds.